At the Instituto de Biomecánica (IBV) we take the scientific knowledge we gain by studying and analysing human functions and turn it into solutions to measure biomechanical variables, thus enabling a variety of problems associated with the study of human motion to be analysed.
These technologies have been adapted to the needs of healthcare professionals involved in managing pathologies affecting the locomotor system, as well as specialists in the evaluation of bodily damage, rehabilitation, traumatology, sports medicine and otorhinolaryngology. Our technologies’ level of versatility and open configuration have made them the gold standard in research fields related to biomechanics in medicine, sports and the workplace.
Our solutions consist of software and a combination of dynamic and kinematic recording techniques that enable a complete evaluation laboratory to be set up.

Our biomechanical evaluation applications are the result of combining protocols for measurements, biomechanical variables that classify the population as normal or pathological, expert information analysis systems, and indices made up of clinical evaluation criteria.
Our organisation is licensed by AEMPS as a manufacturer of medical devices with no. 5098-PS for the products NedLumbar/IBV, NedMano/IBV, NedCervical/IBV, NedRodilla/IBV, NedHombro/IBV, NedCodo/IBV, NedSVE/IBV and NedAMHPlus/IBV.

NedLumbar/IBV
Software to assist specialists in evaluating lower back pain.
NedMano/IBV
Software to assist specialists with evaluations in studying three of the functions of the hand: gripping, distal pinch and lateral pinch
NedCervical/IBV
Software to assist specialists in a functional evaluation of the cervical spine based on a kinematic study of its mobility.
NedRodilla/IBV
Software to assist specialists in a functional evaluation of the knee and lower limb.
NedHombro/IBV
Software to assist specialists in evaluating painful shoulder pathology.
NedCodo/IBV
Software to assist specialists in the evaluation of the elbow joint by recording and analysing various gestures.
NedSVE/IBV
Software to assist specialists in analysing, re-educating and monitoring balance disorders with easily understandable graphic results.
NedLabor/IBV
Software to implement a method for evaluating work capacity that enables possible discrepancies to be identified objectively between a job’s physical requirements and the worker’s physical capacities.
NedAMHPlus/IBV
Software to assist specialists in evaluating the capacity and regularity of gait by comparing it with normal patterns.
Kinemov/IBV
Agile and simple software to analyse agile and simple human motions. Based on open protocols and especially designed for research and analysis of human functions.
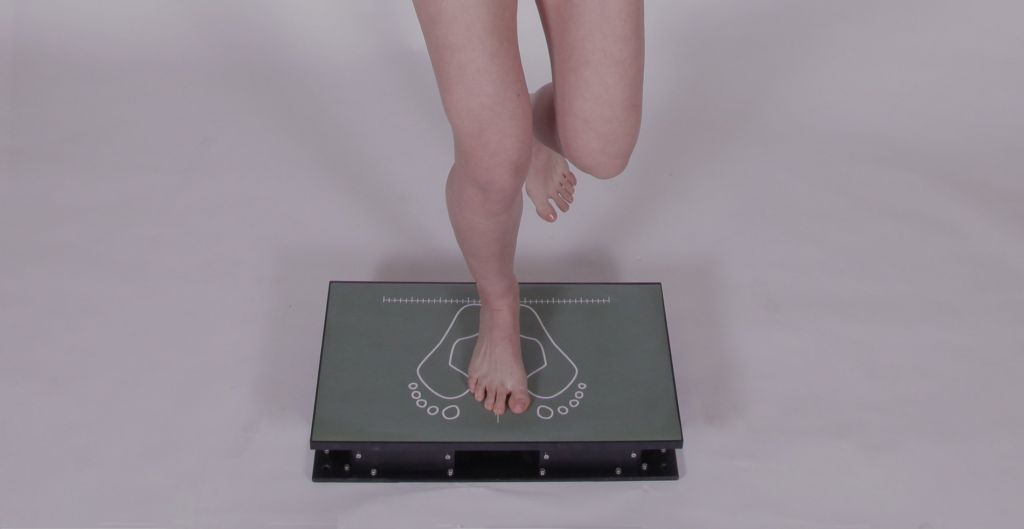
The recording techniques are based on three-dimensional photogrammetry, dynamometric platforms and other instrumental techniques.
Dinascan/IBV
Measurement system based on dynamometric platforms, designed to record and analyse the reaction forces and motions made by a patient on the floor during any type of human activity. The Dinascan/IBV system is available in the P600 and Z14 models.
Kinescan/IBV
A system to analyse motions automatically and in real time, especially designed to record human motions and carry out a kinematic analysis of them.
NedVEP/IBV
Hand dynamometer that records muscle strength and loss of strength associated with the gestures of gripping, lateral pinch and distal pinch.
NedDFM/IBV
Universal dynamometer that records the muscle strength and loss of strength of the different muscle groups in the upper and lower limb.
NedMCV/IBV
Miniature electronic dual inclinometry system for recording mobility limitations and ankylosis of the cervical, dorsal and lumbar regions of the spine.
NedSGE/IBV
Multifunctional electronic goniometry system to record the ranges of motion for the joints of the fingers, wrist, elbow, shoulder, toes, ankle, knee and hip.
IMU/IBV
Inertial sensors system to analyse motions automatically and in real time.

