We generate new knowledge and cutting-edge technologies to be applied to new devices for rehabilitation and personal autonomy.
We are specialists in biomechanics, anthropometry, ergonomics, thermal comfort, UX, physiological signals and the evaluation of cognitive load.
We tackle the challenges in this industry by co-developing projects for companies in collaboration with prescribers, users and other stakeholders. The interaction among companies, technicians and users ensures we create successful solutions.

We provide companies with answers about their products or potential developments for rehabilitation and personal autonomy, concentrating on their interaction with the users and the effect these solutions have on them.
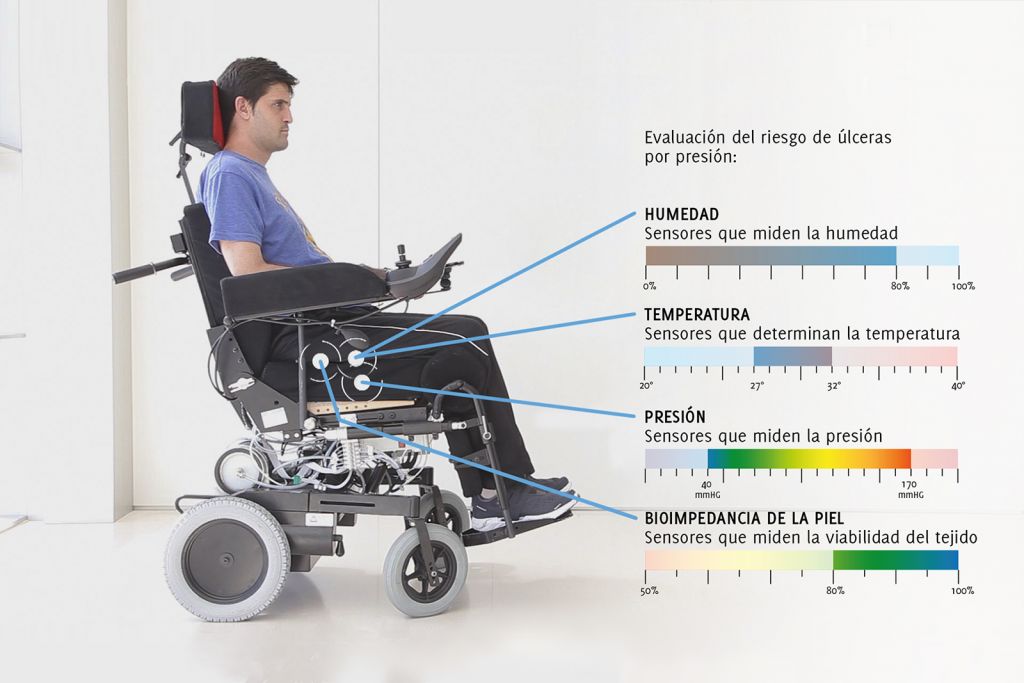
We work with all kinds of products, from classic solutions such as wheelchairs to technological devices such as exoskeletons and tele-rehabilitation systems.
With a multidisciplinary team of researchers and designers, we can take part in any phase of development for a product, providing our know-how in detecting needs, UX, design, prototypes, modelling, simulation and evaluation of medical devices.
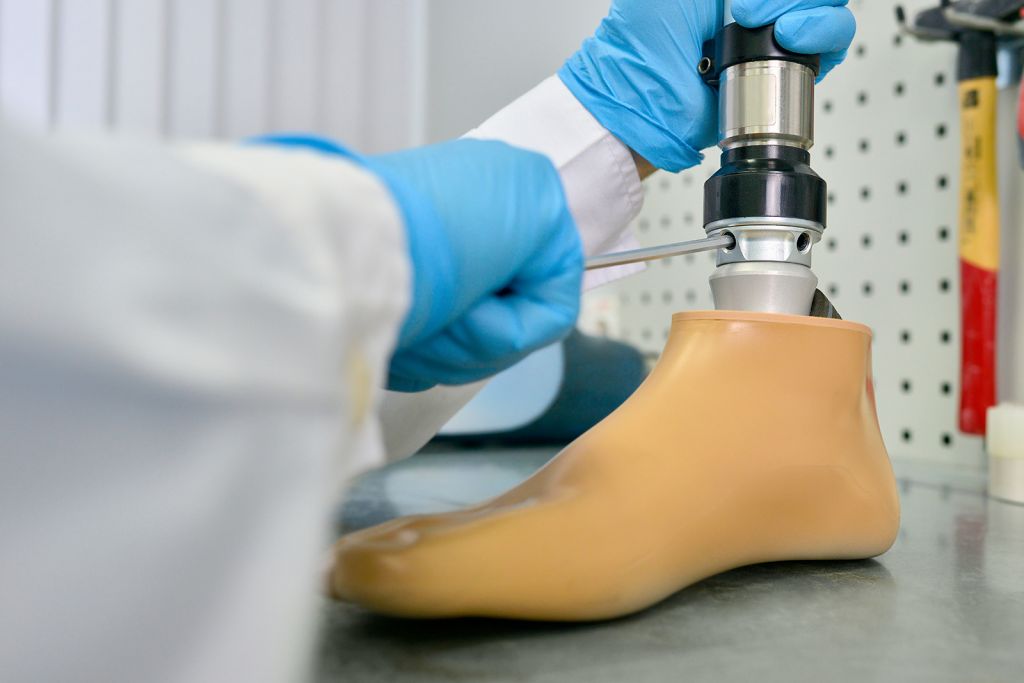
We provide support for all kinds of certification processes for medical devices to attain the FDA and CE markings in: risk analysis, staying abreast of the state-of-the-art, device testing, clinical analysis, software evaluation, etc.
We carry out regulatory testing as well as developing our own protocols so we can assess the devices’ conformity when there is no benchmark regulation available. We can evaluate the devices’ mechanical, ergonomic, thermal and biomechanical behaviour so as to assess almost any of their characteristics

We provide training related to biomechanics, disability, rehabilitation and personal autonomy for professionals and companies.
Together with a multidisciplinary team, we give training programmes on certain topics or else tailored to our clients’ needs.
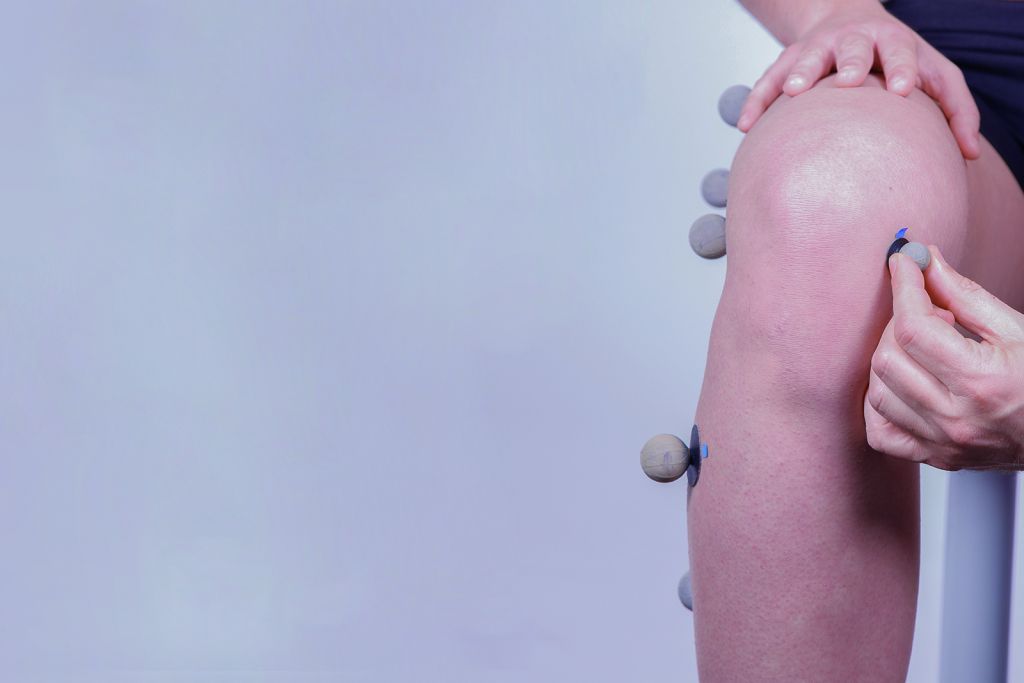
We use biomechanical evaluation to learn the functional capacity of people with disabilities, and the effect of different technological solutions on functions such as gait, balance, coordination, etc. These techniques are commonly used in the process of adapting prostheses and ortheses, and in evaluating the effect of treatment and assistive technology on the locomotor system.