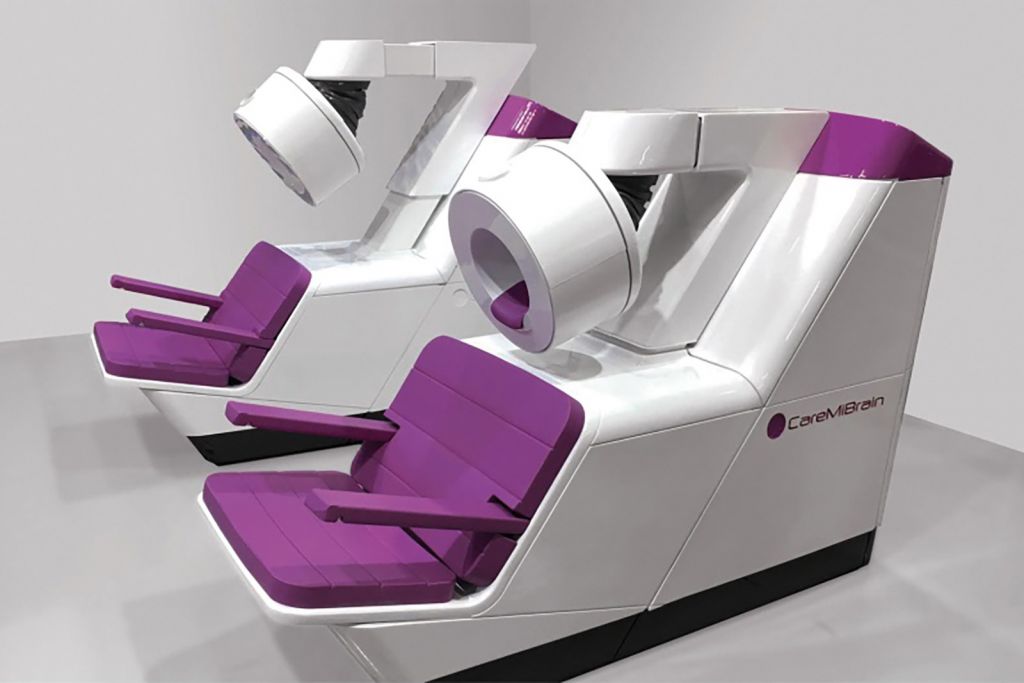
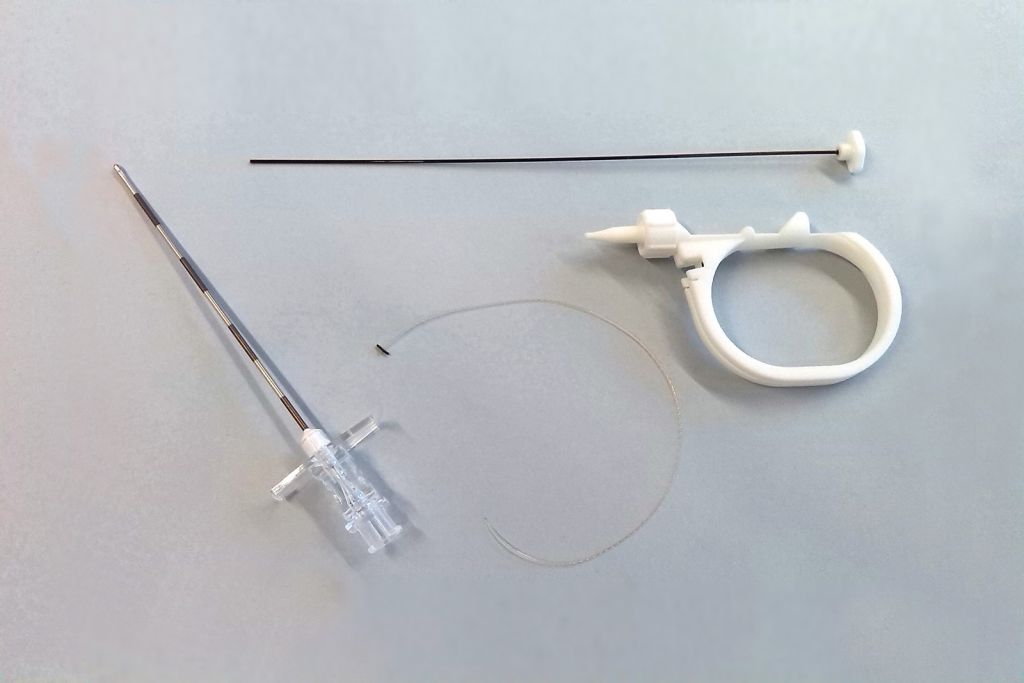
We carry out research to design and develop customised, innovative products for companies so as to improve patients’ quality of life and give a solution to medical specialists’ problems.
Our main projects focus on implants for orthopaedic, dental and veterinary surgery, surgical and laparoscopic instruments for MIS techniques and made-to-measure design, as well as ophthalmologic and cardiovascular applications.
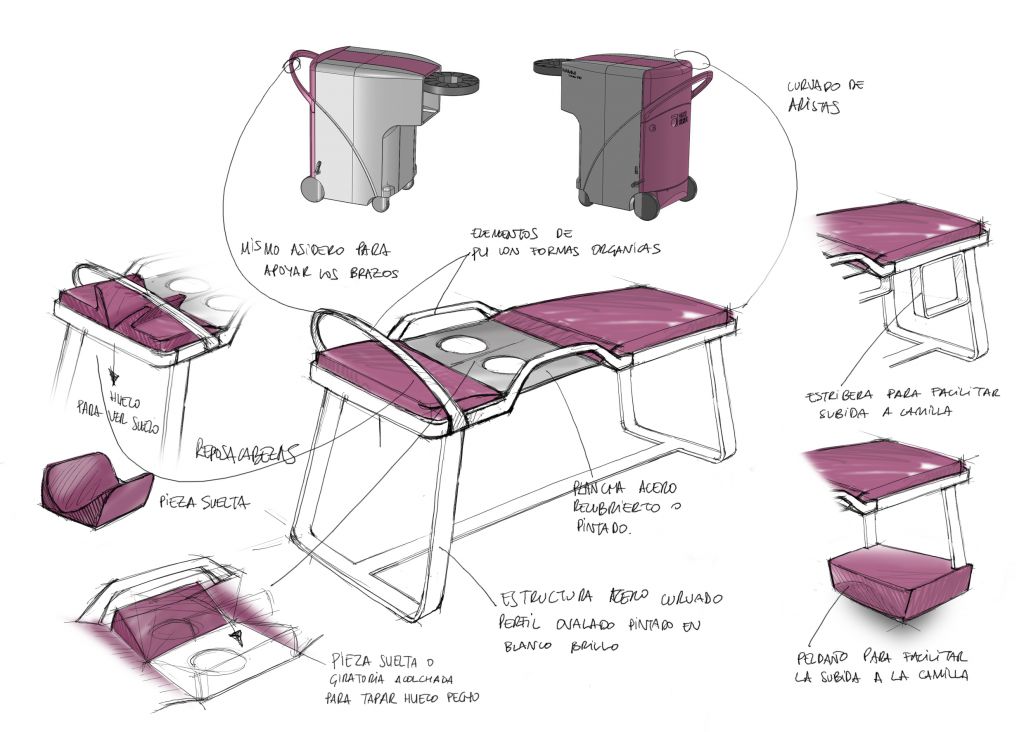
Based on our knowledge about design, biomechanics of body systems, human functions and the way the industry is regulated and works, we provide comprehensive consultancy during the medical device’s design and life cycle, with a modular turnkey service to launch the medical device on the market, adding value and making a real difference to our client.
We carry out consultancy projects in:
– designing and developing products in all their stages.
– assessment about biomaterials.
– providing comprehensive advice as regards the demands and requirements of new regulations on medical devices.

As a referent organisation in the biomechanical evaluation of devices, the Instituto de Biomecánica (IBV) carries out:
Evaluation of medical devices using analytical models (MEF and parametric models).
Evaluation of medical devices via biomechanical testing (static, dynamic, wear resistance and non-regulatory).
Evaluation of biomaterials. Mechanical characterisation, evaluation of toxicity, in vivo evaluation of functioning via implants in animals using different and complementary techniques (densitometric analysis and histomorphometric evaluation).
Together with a multidisciplinary team of experts in biomechanics, design, biomaterials and healthcare, we provide training services through specific programmes and education solutions tailored to each client’s needs in aspects related to biomechanics and healthcare technologies.

Personalisation is one of the great challenges in the medical devices industry. The evolution of processes and technologies for 3D scanning and modelling by using anthropometric data from different segments of the population enables precise, high-resolution reconstruction of the human body and its parts. High resolution 3D technologies allow for applications in industries and disciplines such as aesthetic medicine, plastic surgery, cosmetics and personal care products, and more
3D face scanner
High resolution scanner enabling reconstruction of the face’s 3D geometry and texture
3D face scanner
High resolution scanner enabling reconstruction of the face’s 3D geometry and texture